Energy of Electron in Nth Bohr Orbit-
Before you go through this article, make sure that you have gone through the previous article on Bohr’s Atomic Model.
We have learnt that-
- Electrons revolve around the nucleus in fixed energy orbits called as stationary states.
- The centripetal force required for their revolution is provided by the electrostatic force of attraction between the nucleus and the electron.
In this article, we will derive the expression for the energy of electron in nth Bohr orbit.
Kinds of Energies-
Consider a single electron species having atomic number Z. It consists of a nucleus with a charge +Ze and a single electron of charge -e which revolves around it in a circular orbit of radius R with a speed v.
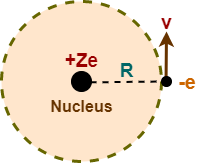
The following two kinds of energies are associated with an electron-
- Kinetic Energy
- Electrostatic Potential Energy (of the two charges)
Kinetic Energy of Electron in Nth Bohr Orbit-
Due to its motion (revolution) around the nucleus, an electron possess kinetic energy.
The kinetic energy of the electron in nth Bohr orbit is given by-

Also Read- Speed of Electron in Nth Bohr Orbit
Potential Energy of Electron in Nth Bohr Orbit-
The electrostatic potential energy of the electron in nth Bohr orbit is given by-

Also Read- Radius of Nth Bohr Orbit
Total Energy of Electron in Nth Bohr Orbit-
The total energy of the electron in nth Bohr orbit is given by-
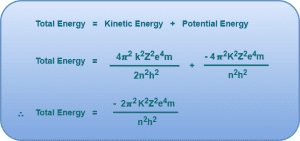
This is the required expression for the total energy of electron in nth Bohr orbit.
Important NoteIt is important to note that the total energy of electron is negative. The negative value of the total energy indicates that the electron is bound to the nucleus by means of electrostatic attraction and some work is required to be done to pull it away from the nucleus. |
Relation Between Kinetic Energy, Potential Energy and Total Energy of Electron-
From the above expressions of different energies, we can infer the following relationship between kinetic energy, potential energy and total energy of electron in the nth orbit-
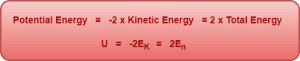
Trick To Learn-
You are too negative of Kinetic energy and twice of total energy.
Simplified Expression For Total Energy of Electron in Nth Bohr Orbit-

This is the simplified expression for the total energy of electron in nth Bohr orbit.
For hydrogen atom (Z=1),
- The total energy of electron in 1st Bohr orbit is E1 = -13.6/12 = -13.6 eV
- The total energy of electron in 2nd Bohr orbit is E2 = -13.6/22 = -3.4 eV
- The total energy of electron in 3rd Bohr orbit is E3 = -13.6/32 = -1.51 eV
- The total energy of electron in 4th Bohr orbit is E4 = -13.6/42 = -0.85 eV
- The total energy of electron in 5th Bohr orbit is E5 = -13.6/52 = -0.54 eV
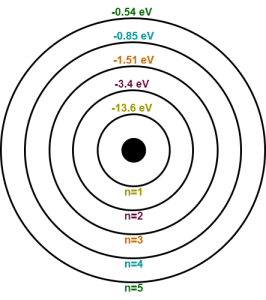
It is very important to memorize these values of total energies for solving numerical problems with ease.
Read the next article on-
Spectral Series of Hydrogen Atom
Get more notes & other study material of the Chapter Atoms.

